  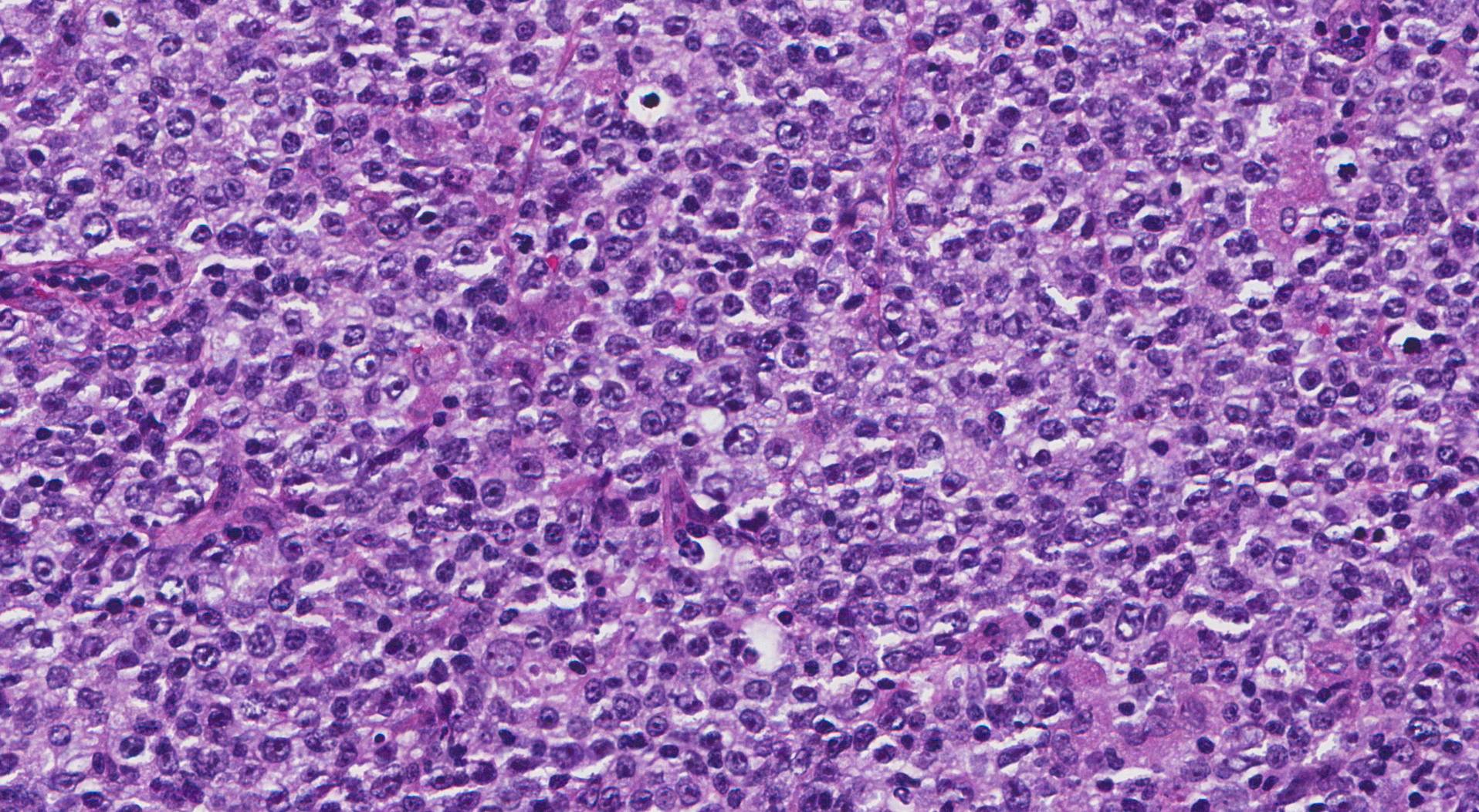
In addition, ALK dysregulation is not unique to ALCL and has been detected in inflammatory myofibroblastic tumors, carcinoma, tumors of neural origin, and in the peripheral blood cells of healthy people by sensitive, nested reverse transcription–polymerase chain reaction. In particular, recognition of ALCL with variant histology is difficult, and distinction of C-ALCL from lymphomatoid papulosis (LyP) and from systemic ALCL may be challenging. 17– 19 Although ALK+ ALCL and C-ALCL are well characterized, diagnostic pitfalls remain. 10– 16Ĭurrently, 3 distinct T-cell tumors (ALK+ ALCL, ALK− ALCL, and primary cutaneous ) are described in the 2008 World Health Organization (WHO) classification. After widespread immunohistochemical analysis with anti-ALK antibodies, 8, 9 ALK+ ALCL was defined as a specific entity that typically affects children and young adults, has a morphologic spectrum that includes small cell and lymphohistiocytic variants, and in most cases has a better prognosis than ALK− ALCL. Because ALK is not normally expressed in lymphoid tissue, anti-ALK antibodies can be used as a surrogate method for detecting t(2 5). 3– 6 In 1994, the translocation was cloned by Steve Morris and others 7 at St Jude Children's Research Hospital in Memphis, Tennessee, and was found to involve a receptor tyrosine kinase called anaplastic lymphoma kinase ( ALK) on 2p23 and nucleophosmin ( NPM) on 5q35. In the late 1980s and early 1990s, a recurrent chromosomal translocation t(2 5) was described. The rather broad expression of CD30 in reactive processes and other tumors led many individuals to question whether ALCL was a distinct entity. 2 Subsequently, Ki-1 was identified as an activation antigen (now designated CD30) and a member of the tumor necrosis factor receptor family. In 1985, Harald Stein and Karl Lennert with colleagues 1 identified a unique large cell lymphoma with anaplastic cytology, an unusual sinus growth pattern, and strong expression of the antigen Ki-1 recognized by an antibody developed in Kiel, West Germany. Nevertheless, pathomechanistic, therapeutic, and diagnostic challenges remain that should be resolved as we embark on the next generation of discovery. Constitutive NPM-ALK signaling has been shown to drive oncogenesis via an intricate network of redundant and interacting pathways that regulate cell proliferation, cell fate, and cytoskeletal modeling. Major strides have been made during the last decade in our understanding of the complex pathogenesis of ALCL. Particular emphasis is given to the biology and pathogenesis of ALCL.ĭata Sources-Search of the medical literature (Ovid MEDLINE In-Process & Other Non-Indexed Citations and Ovid MEDLINE: 1950 to Present ) and more than 20 years of diagnostic experience were used as the source of data for review.Ĭonclusions-Based on immunostaining for activation antigen CD30 and the presence of dysregulation of the anaplastic lymphoma kinase gene (2p23), the diagnosis of ALCL has become relatively straightforward for most patients. Objective-To summarize our current knowledge of the clinical and pathologic features of systemic and primary cutaneous ALCL. Investigators at many universities and hospitals worldwide have contributed to our current in-depth understanding of this unique peripheral T-cell lymphoma, which in its systemic form, principally occurs in children and young adults. Context-The year 2010 commemorates the 25th year since the seminal publication by Karl Lennert and Harald Stein and others in Kiel, West Germany, describing an unusual large cell lymphoma now known as anaplastic large cell lymphoma (ALCL). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
